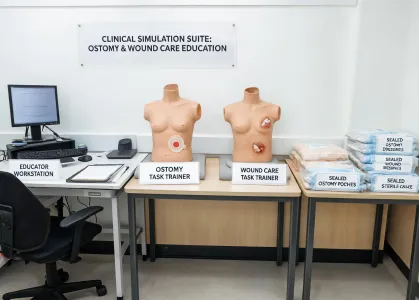
Clinical Simulation Suite
Education scenarios help teams standardize application technique and teach-back routines.
Evidence-led chronic care programs for wound, ostomy, skin, and continence teams that need defensible claims and reliable supplier documentation.
"Every product education claim must be clear enough for a nurse educator, a value analysis committee, and a patient support team to interpret the same way."Clinical Governance Lead, Coloplast
Coloplast is presented here as an authority-oriented medical consumables partner focused on chronic care pathways. The site emphasizes clinical documentation, IFU clarity, user education, procurement evidence, and post-market feedback discipline because these are the areas that hospital buyers repeatedly inspect when consumables move from trial to standard formulary. Rather than positioning the portfolio as a loose set of SKUs, the content groups wound dressings, skin protection, ostomy support, and continence care around the documentation that healthcare systems need to keep care transitions stable.
The brand voice is deliberately measured. It avoids exaggerated claims and instead explains how clinical use instructions, training records, product substitution notes, and traceability materials can be reviewed by nursing leadership, quality managers, supply chain teams, and home health coordinators. This makes the site useful for cross-functional purchasing conversations where each stakeholder asks a different question about the same product family.

Education scenarios help teams standardize application technique and teach-back routines.
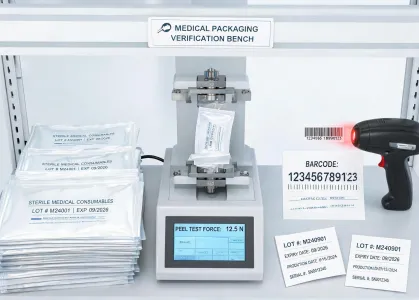
Packaging configuration, label readability, and handling instructions support procurement review.

Documentation focuses on skin protection, wear time considerations, and material compatibility.
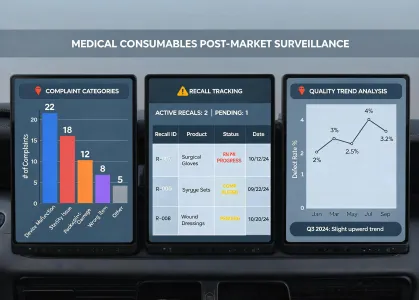
Feedback intake, trend review, and escalation routing remain visible after adoption.
Get a Coloplast documentation packet built for clinical leadership, supply chain, quality, and patient support review.
Request Dossier